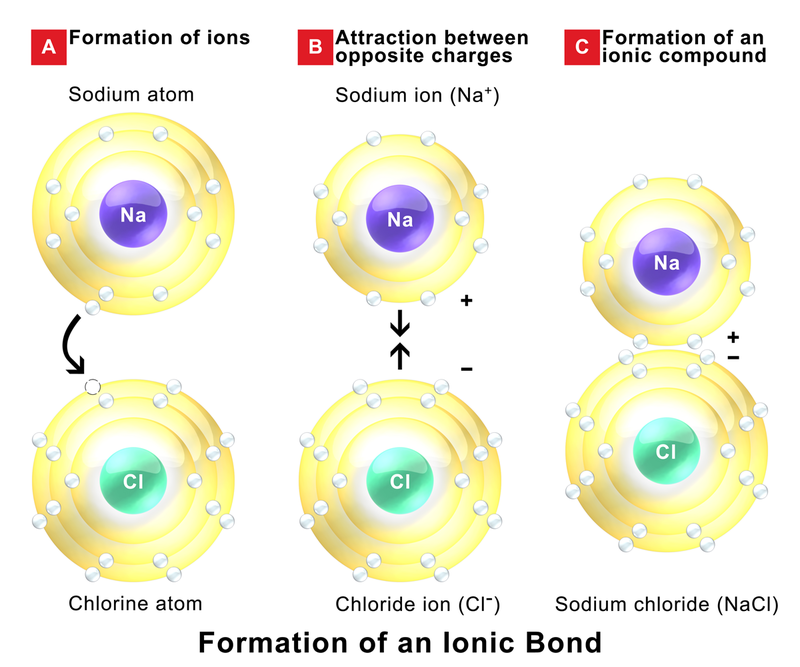
Bonds between a non-metal and a metal are ionic (For example, a Na +Cl - bond is ionic.(For example, a P-S bond is a covalent bond.) Bonds amongst non-metal atoms are covalent.Later, you will be able to determine just how ionic or covalent a bond will be, but for now here are some guidelines to follow: Metallic Bonds - occurs in metals (similar to covalent bonds).(1-3) However, the functions of many metal ion-binding proteins remain unclear. The classical type of complex has a central metal ion surrounded by coordinating ligands, with little electronic delocalization between them, such that the ligand metal bonding is essentially ionic. Electrostatic (Ionic) Bonds - cations and anions are held together by electrostatic attractions There are many different types of metal ion-binding proteins, and those that bind the most common metal ions, such as iron, usually regulate essential functions in physiological processes.Covalent Bonds - atoms are held together by sharing electrons.They arise from a difference in the electronegativities of the bonded atoms. Later will we examine the energetic and thermodynamic bases of this personification.) Ionic bonds involve the transfer of electrons between two chemical species. (It is quite common for chemists to personify the atoms and molecule with which they work! Saying an atom wants an another electron is akin to saying a ball wants to roll down a hill. The desire of atoms to gain or lose electrons to get a noble gas number of electrons is what leads to chemical bonding. They have the optimum number of electrons and don't like to form chemical bonds. Only the noble gases (column 8A) are content with the number of electrons. They are formed because atoms are not happy with the number of electrons that they have. Chemical bonds are the forces that hold atoms together in compounds.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
